Introduction
Dispersal (expansion of individuals) is the most significant process supporting genetic diversity and the population size of birds (Greenwood, 1980). The distance of dispersal depends on sex, age, success of previous reproduction and a number of other population parameters (Greenwood, Harvey, 1982; Sokolov, 1991; Haas, 1998; Pardis et al., 1998). In most cases the dispersal of young birds (natal dispersal) exceeds that of adult birds (breedng dispersal), at that the dispersal of females is higher than that of males. The distribution of actual values of natal and breedng dispersal is often bimodal (Pakanen et al., 2011). It enables to conditionally separate short dispersal (displacement of an individual within area of its birth and previous reproduction) and long dispersal (displacement outside this area) (Barrowclough, 1978; Payne, Payne, 1990; Pakanen et al., 2011). The short dispersal is assessed based on movement monitoring of marked individuals. The direct assessment of the long dispersal is complicated in a lot of cases, since it requires the exploration of vast areas (Barrowclough, 1978; Paradis et al., 1998). One can indirectly judge the intensity of long dispersal on the basis of the analysis of the population dynamics (Bursky, 2008) or the apparent survival of birds (Payne, Payne, 1990; Pakanen et al., 2011). The purpose of the paper is to assess the natal and nestling dispersal of the yellow wagtail (Motacilla flava) based on the records of returns of marked individuals and the analysis of the apparent survival in a local population as well as to reveal the relationship between the distance of nesting dispersal and the success of the previous reproduction of the yellow wagtail.
Materials
The investigation was carried out in the southern part of the Russky sever national park (Kirillovsky district, Vologda region) in the large plot of abandoned agricultural land in the vicinity of the village Topornya (59º 46´ N.L.,38º 22´ E.L.) in 2005-2018. The species model (yellow wagtail) is a distant spotty distributed migrant tended to aggregative nesting. At that compact or diffuse local settlements from several pairs to several tens of pairs can be separated by large areas of unoccupied species habitats (Tsvetkov, 2004; Shitikov et al., 2013). During the study in the station there existed 3 diffuse settlements of the yellow wagtail, where the total number of breeding yellow wagtails reached 50 pairs. In two settlements individual marking of adult yellow wagtails (adult birds and nestlings) and monitoring of their successful reproduction was carried out. The search of returned marked birds was produced in the whole station. Considering the isolated location of the control plot among the habitats unsuitable for species nesting, we can completely monitor all the returns to the area of birth (or previous reproduction); not returned birds were considered killed or emigrated (committed long dispersal). In the paper the data of 104 adult yellow wagtails and 406 nestlings were analyzed. For them 13 measurement of natal dispersal and 55 – of breeding one were received.
Methods
Field works were produced annually from the third decade of May till the third decade of July. They included the search of nests, capture and individual color marking of adult yellow wagtails, control of breeding success as well as the records of returns of earlier marked individuals. Found nests were mapped using GPS navigator Garmin 60s. Adult birds were captured near nests using mist net or automatic bird nets 6-7 days after chicks hatching and marked with the individual combination of color rings. A bird was considered successfully breeding if at least one chick had left the nest. Chicks were ringed in the nests 5-9 days after hatching, at that all the chicks of the same brood received the same combination of color rings. Natal dispersal was measured as a distance between the birth nest and the first known own nest, nestling dispersal – as a distance between two nests of the same bird in successive years. If the nest of a returned yellow wagtail was not found, the distance to the centre of its nest territory was measured. Distance measurements were carried out on the basis of geographical coordinates using the Geosphere software module (Hijmans et al., 2014) in R 3.2.3 (R Core Team, 2016).
The apparent survival of young yellow wagtails was determined by means of the stochastic Cormack-Jelly-Seber model in the program MARK 8.0 (White, Burnham, 1999). To assess the survival and re-registration possibility a phi(age)p(age) model was constructed, where both the estimated indicators (apparent survival and re-registration possibility) were considered age dependent. To improve the accuracy of the assessment of these parameters for young birds, the fixed values of the apparent survival (φ = 0.32) and re-registration possibility (p = 1.00) of adult yellow wagtails received earlier for the same local population were used (Shitikov et al., 2017). The models with time-dependent parameters were not tested because our samples of birds marked annually do not allow that. We also did not test the effect of bird sex on the apparent survival, because we could not determine the sex of nestlings during ringing.
The effect of sex and previous breeding success on the breeding dispersal of yellow wagtails was assessed using linear mixed-effect models (LMM). The logged value of breeding dispersal was used as a dependent variable, and sex, individual breeding success and their interaction – as independent variables. The calendar year and the individual number of a bird were considered as random factors. The information-theoretical approach was used to choose the model (Burnham, Anderson, 2002). Models were ranked with the help of informational criterion of Akaike corrected for small samples (AICc). Models possessing the ΔAICc values less than 2 were considered adequately describing data. The simulation was performed in R 3.2.3 (R Core Team, 2016) using the lmr4 module (Bates et al., 2016). Models were ranked using the dredge function of the Mumin module (Bartoń, 2013).
Results
From 406 nestlings ringed in 2005-2017, in consequent years only 13 (return rate – 3.2%) returned, among them there were 11 males and 2 females. The year after birth 11 yellow wagtails were found, 2 more birds were found only in a year. The apparent survival of young birds assessed using the model φ(age)p(age) was φ = 0.04 ± 0.01, and the re-registration possibility p = 0.62 ± 0.18. The median of natal dispersal (n = 13) for all individuals was 2200 m (350-4989 m) that corresponds to the parameter calculated only for males, at that the returned females were found at the distance of 481 and 3304 m from their native nests. The analysis of the distribution of natal dispersal distances (Fig.1) shows that most of the returned young yellow wagtails have nested at a considerable distance (more than 500 m) from their native nests.
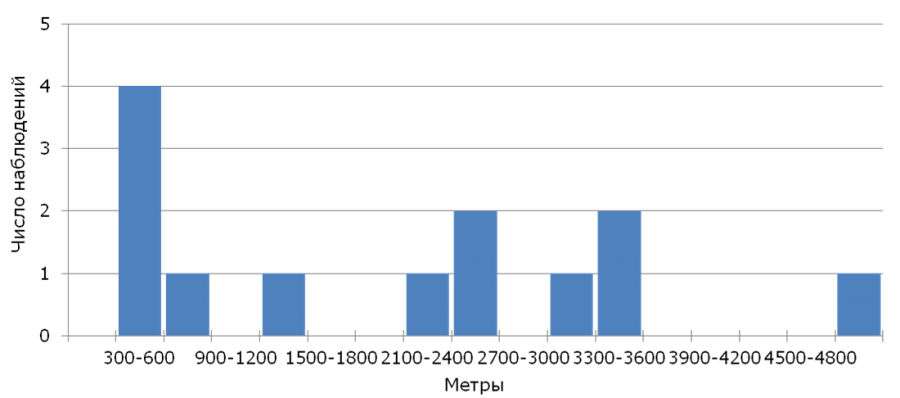
Fig.1. Distribution of breeding dispersal distances of the yellow wagtail
The median of breeding dispersal (n = 55) was 195 m (1-3011 m), for males (n = 28) it was 116 m (12-2922 m), for females (n = 27) – 251 m (1- 3011 m). The vast majority of both sex adult yellow wagtails were found within a radius of 300 m from their previous year’s nests (Fig.2).
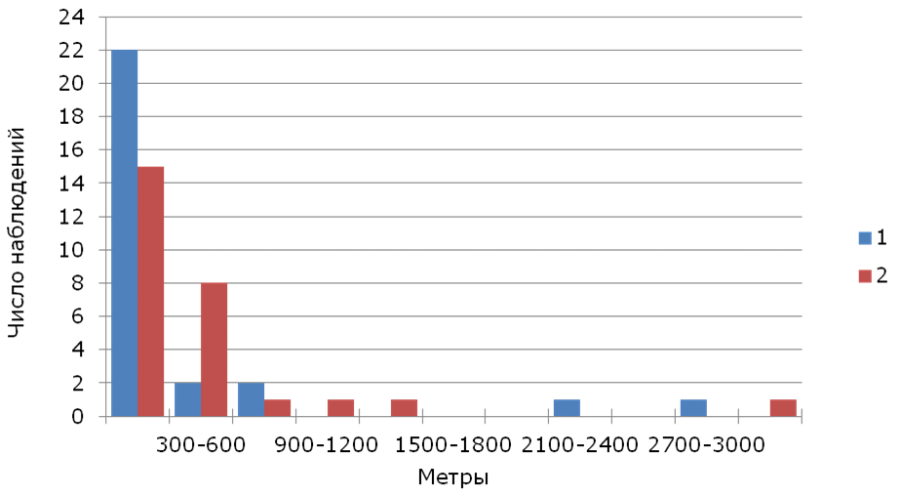
Fig.2. Distribution of breeding dispersal distances of yellow wagtails. 1 – males, 2 – females
Returned, successfully breeding yellow wagtails (n = 33) built nests 195 m (1-2992 m) from their previous year’s nests, while the median of breeding dispersal of unsuccessfully breeding birds (n = 22) was 212 m (1-3011 m). The simulation results show that the sex of the returned wagtails and their individual breeding success had no effect on the amount of breeding dispersal, since only a contact model possesses the values ΔAIC less than 2 units (Table 1). All the models taking into account the factors “Sex” and “Success”, and their combinations had ΔAICc values of more than 2 units..
Table1. Ranking models of the effect of sex and breeding success on the value of yellow Wagtail breeding dispersal in the Russky Sever national park.
| Model | df | ΔAICc | Model weight |
| const | 4 | 0.00 | 0.74 |
| Sex | 5 | 3.65 | 0.12 |
| Success | 5 | 3.72 | 0.12 |
| Sex+Success | 6 | 7.47 | 0.02 |
| Sex+Success+Sex*Success | 7 | 9.90 | 0.01 |
Discussion
The apparent survival of young yellow wagtails in the Russky Sever national park was extremely low. The estimates of survival received by the stochastic Cormack-Jolly-Seber model (4 ± 1 %) are located near the lower limits (0-13.5%) known for the small Passeriformes – long-distance migrants (Sokolov, 1991; Weatherhead, Forbes, 1994; Maness, Anderson, 2013). Unfortunately, the estimates of the survival of young yellow wagtails in other parts of the species area are lacking in literature. The estimates of the apparent survival of juveniles (2-4%) similar to ours were obtained in Alaska for the close species – Eastern Yellow Wagtail Motacilla (flava) tchutchensis (Renner, McCaffery, 2008). Our results adjusted for the re-registration probability of marked birds showed that 96% of young wagtails did not return to the birth area. We assume that the considerable part of them survived, but emigrated outside their birth area (i.e. committed long dispersal), since with a mortality rate of about 96% in the first year of life, any local population would be doomed to rapid extinction. The information about the range of such dispersal is very scarce. Earlier we managed to find the male of the yellow wagtail nesting at a distance of 14 km from its native nest (Shitikov et al., 2013). According to the analysis of the return of rings from dead birds (n = 28), in the Great Britain the average natal dispersal of the yellow wagtail was 12.5 km (Paradis et al., 1998). A few yellow wagtails returning to their birth area did not seek to occupy the territory near their native nests, as it is evidenced by the distribution of natal dispersal distances. Besides, it should be noted that the returns of young females are almost completely absent, this confirms the generally accepted view of less fidelity to the area of birth in young females compared to males.
The apparent survival of adult yellow wagtails in the studied local population depended on the previous breeding success and was 0.39 ± 0.06 for successfully breeding birds, but it was 0.19 ± 0.06 for unsuccessfully breeding ones (Shitikov et al., 2017). Thus, the majority of successfully nesting yellow wagtails returned to the area of their previous breeding, while a considerable part of unsuccessfully breeding birds left it (i.e., committed long dispersal). Nevertheless, the apparent survival of unsuccessfully breeding adult birds significantly exceeded that of young birds that once again testifies to high aspiration of the last to leave the area of their birth. Significant differences in the survival of successfully and unsuccessfully breeding individuals are often connected with the difference in the breeding dispersal of the two groups: unsuccessfully breedng birds shift to a greater distance in comparison with successfully breeding ones (Haas, 1998; Hoover, 2003; Sedgwick, 2004). Our data only partially confirm this provision: the previous breeding success affected only the loyalty to the nesting area (i.e., the probability of committing long dispersal). At that, the yellow wagtails that returned to the nestling area chose the territory regardless of the previous breeding success. Moreover, most of the returned wagtails (regardless of the success of the previous breeding) build nests at a distance of less than 300 m from their previous nests, i.e., in fact, the birds sought to occupy their last year's nestling.
Conclusions
Connectivity between the local populations of the yellow wagtail in European North of Russia is maintained mainly due to natal dispersal. Most young birds leave their birth area forever, a few returned birds shift to the considerable distances relative to their native nests. At that, the basis of nesting settlements is made up of previously successfully breeding individuals who seek to occupy nestling areas in close proximity to those occupied in previous years.
References
Barrowclough G. F. Sampling bias in dispersal studies based on finite area, Bird-banding. 1978. Vol. 49. P. 333–341.
Bartoń K. MuMIn: multi-model inference, R package version 1.9.5. 2013. URL: http://CRAN.R-project.org/package=MuMIn.
Bates D., Mächler M., Bolker B., Walker S. lme4: linear mixed-effects models using Eigen and S4, R package version 1.1-12. 2016. URL: https://CRAN.R-project.org/package=lme4.
Burnham K. P., Anderson D. R. Model Selection and Multimodel Inference: A Practical Information Theoretic Approach. Springer, New York. 2002.
Burskiy O. V. Habitat selection and metapopulation structure: a multi-year study of distribution of the Hodgson's pipit, Anthus Hodgsoni Richm. (Aves, Passeriformes), Zhur. obsch. biol. 2008. T. 69. Vyp. 5. P. 323–343.
Cvetkov A. V. Aggregated settlements of yellow and white wagtails (Motacilla flava, M. alba), Rup. ornit. zhurn. 2004. T. 3. P. 227.
Greenwood P. J. Mating systems, philopatry and dispersal in birds and mammals, Anim. Behav. 1980. Vol. 28. P. 1140–1162. doi: 10.1016/s0003-3472(80)80103-5.
Greenwood P. J., Harvey P. H. The natal and breeding dispersal of birds, Annu. Rev. Ecol. Syst. 1982. Vol. 13. P. 1–21.
Haas C. A. Effects of Prior Nesting Success on Site Fidelity and Breeding Dispersal: An Experimental Approach, Auk. 1998. Vol. 115. P. 929–936.
Hijmans R., Williams E., Vennes C. Geosphere: spherical trigonometry, R package version 1.3-11. 2014. URL: http://CRAN.R–project.org/package=geosphere.
Hoover J. P. 0416:drfsfi2.0.co;2.
Maness T. J., Anderson D. J. Predictors of juvenile survival in birds, Ornithological Monographs. 2013. Vol. 78. No 1. P. 1-55. doi: 10.1525/om.2013.78.1.1.
Pakanen V, M., Hildén O., Rönkä A., Belda E. J., Luukkonen A., Kvist L., Koivula K. Breeding dispersal strategies following reproductive failure explain low apparent survival of immigrant Temminck’s stints, Oikos. 2011. Vol. 120. P. 615–622. doi: 10.1111/j.1600-0706.2010.18953.x.
Paradis E., Baillie S. R., Sutherland W. J., Gregory R. D. Patterns of natal and breeding dispersal in birds, J. Anim. Ecol. 1998. Vol. 67. P. 518–536.
Payne R., Payne L. Survival estimates of Indigo Buntings: comparison of banding recoveries and local observation, Condor. 1990. Vol. 92. P. 938–946.
R Core Team. R: A language and environment for statistical computing, Vienna: R Foundation for Statistical Computing. 2016. URL: https://www.R-project.org.
Renner H. M., McCaffery B. J. Demography of Eastern Yellow Wagtails at Cape Romanzof, Alaska, Wilson J. Ornith. 2008. Vol. 120. P. 85–91. doi: 10.1676/06-122.1
Sedgwick J. A. 1103:sftfan2.0.co;2.
Shitikov D. A. Vaytina T. M. Makarova T. V. Fedotova S. E. Krasnyh N. A. Yurchenko Yu. A. Breeding success effect on the apparent survival of grassland passerines, Zool. zhurn. 2017. T. 96. No. 7. P. 827–837.
Shitikov D. A., Dubkova E. V., Makarova T. V. The demography of Yellow Wagtails Motacilla flava on abandoned fields in northern European Russia, Bird Study. 2013. Vol. 60. P. 518–526. doi: 10.1080/00063657.2013.850467.
Sokolov L. V. Philopatry and dispersal in birds, Trudy Zool. in-ta AN SSSR. 1991. Vyp. 230. P. 1–232.
Weatherhead P. J., Forbes M. R. L. Natal philopatry in passerine birds: genetic or ecological influences?, Behav. Ecol. 1994. Vol. 5. P. 426–433. doi: 10.1093/beheco/5.4.426.
White G. C., Burnham K. P. Program MARK: survival estimation from populations of marked animals, Bird Study. 1999. Vol. 46. P. 120–139.





 © 2011 - 2025
© 2011 - 2025